CARAT - Chimeric Antigen Receptors (CARs) for Advanced Therapies
an EU-funded collaborative research project involving 8 partners from 4 different European countries (France, Germany, Italy & United Kingdom).
- CARAT's goal
is to develop an efficient and safe technology platform for advanced cellular therapies, namely to manufacture CAR T-cell products for personalized treatment of cancer patients.
- Recent Success Stories
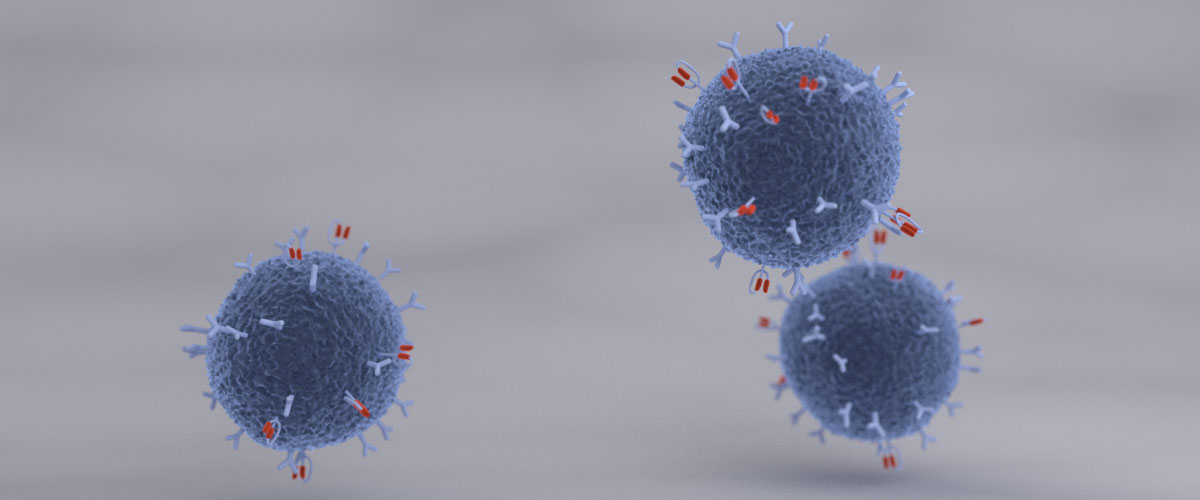
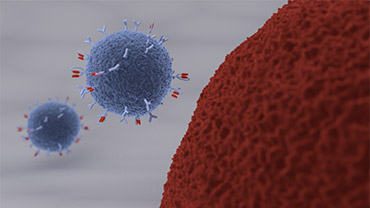
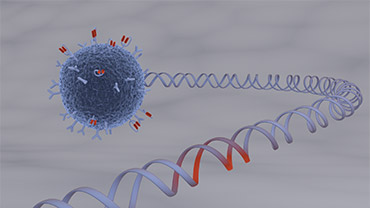
of CAR-based therapies have raised enormous expectations to cure severely ill patients: CARs were introduced into cells of the immune system, typically T-cells, by genetic engineering.
- Initial clinical studies
demonstrated considerable evidence that these artificial recognition molecules allow specific targeting of immune responses towards complete and stable eradication of cancer cells.
- Translation into the clinic
of such advanced therapeutic concepts is extremely difficult as the manufacture of CAR-modified T-cells is technologically complex and expensive.
- The CARAT technology platform
will thus enable the automated, safe, and cost efficient manufacture of more effective CAR-modified T-cells. Thereby, CARAT will spread the clinical use of CAR T-cell technologies with the ultimate goal of serving patients with so far incurable hematologic malignancies and solid tumours like colon, pancreas or lung cancer – everywhere in Europe.
CARAT Network
The CARAT consortium comprises a multi-national team of leading experts from eight European partnering institutions.
Benefits
Recent success stories of cancer therapy with CAR-modified T-cells have raised enormous scientific and public expectations to cure severely ill patients.
Latest News
-
Landscape study on patent filing – CAR T-cell Immunotherapy
This study commissioned by the European Patent Office demonstrates the application of patent information to gain insights into specific technological... -
CARAT Fellowship Programme 2019: Seven exceptional scientists benefit from unique know-how
To enable more and more experts to use innovative cell manufacturing technology: this is the goal of the CARAT Training Fellowship Programme. In...
Publications
-
Tumor-Specific Delivery of Immune Checkpoint Inhibitors by Engineered AAV Vectors.
Front Oncol 2019 Feb 14;9:52. doi: 10.3389/fonc.2019.00052. eCollection 2019.
-
Highly Efficient and Selective CAR-Gene Transfer Using CD4- and CD8-Targeted Lentiviral Vectors.
Mol Ther Methods Clin Dev. 2019 Mar 16;13:371-379. doi: 10.1016/j.omtm.2019.03.003. eCollection 2019 Jun 14.
-
In vivo generation of human CD19‐CAR T cells results in B‐cell depletion and signs of cytokine release syndrome
EMBO Mol Med Sep 17 [Epub ahead of print]